|
April 3, 2020 |
ARUP Endorses ACLA’s Appeal to CDC for Help Educating Clinicians The ACLA has asked the CDC to take immediate steps to educate healthcare providers and the public about the importance of prioritizing COVID-19 |
|
April 3, 2020 |
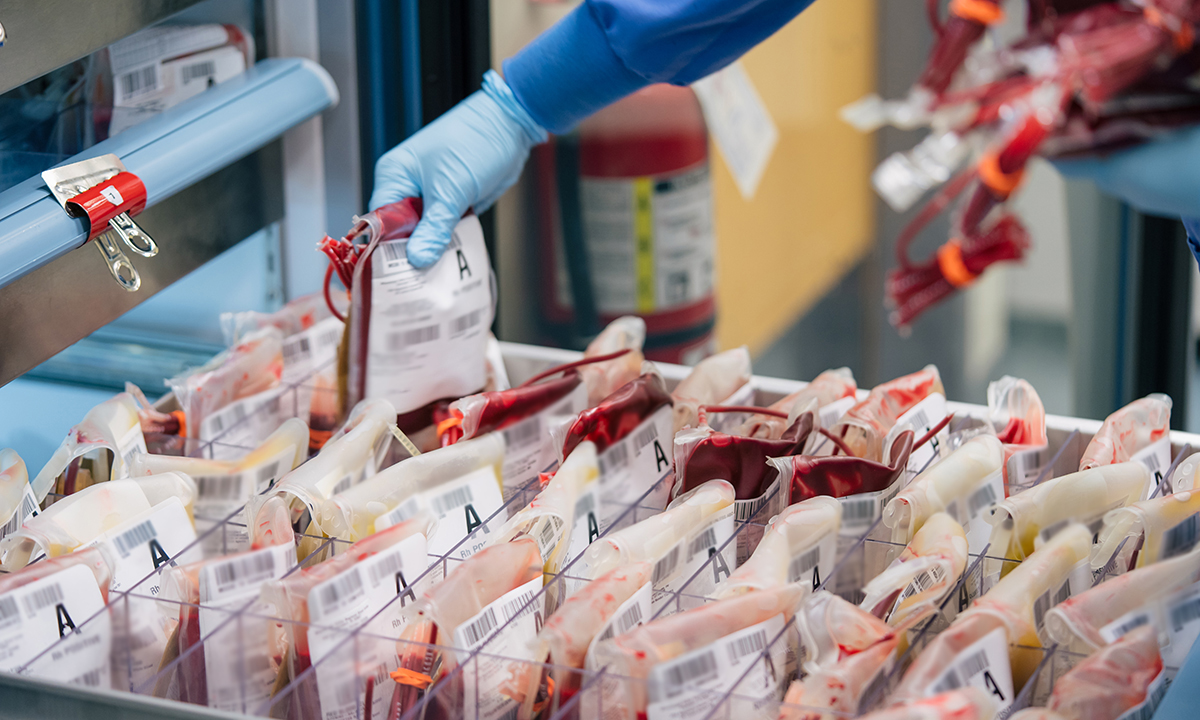
While most blood drives have been canceled, ARUP’s two blood donation centers have remained open. ARUP has seen donations increase as more people have sought ways |
|
March 31, 2020 |
ARUP and Intermountain Healthcare Collaborate to Meet COVID-19 Testing Needs As the state of Utah continues to expand testing to halt the spread of COVID-19, its two largest laboratory services providers |
|
March 16, 2020 |
Due to supply constraints in the face of extraordinary demand for COVID-19 testing, ARUP told its national clients it can no longer accept orders for its 2019 Novel |
|
March 10, 2020 |
ARUP Laboratories today announced the launch of its new test for the novel coronavirus. ARUP will start COVID-19 testing on March 12, 2020. |
|
February 20, 2020 |
FDA Files ARUP Laboratories’ AAV5 CDx for Hemophilia A Gene Therapy Treatment ARUP Laboratories announced today that the Food and Drug Administration (FDA) has filed ARUP’s premarket approval |