ARUP’s award-winning returnship program helps experienced professionals relaunch their careers while strengthening the life sciences workforce.
Join ARUP in celebrating Lab Week with stories that highlight the many ways laboratory professionals go above and beyond to provide excellent patient care.
Connect with ARUP experts to learn about our comprehensive neurology testing suites and how intentional lab testing can support clinically meaningful insights across a range of neurologic conditions.
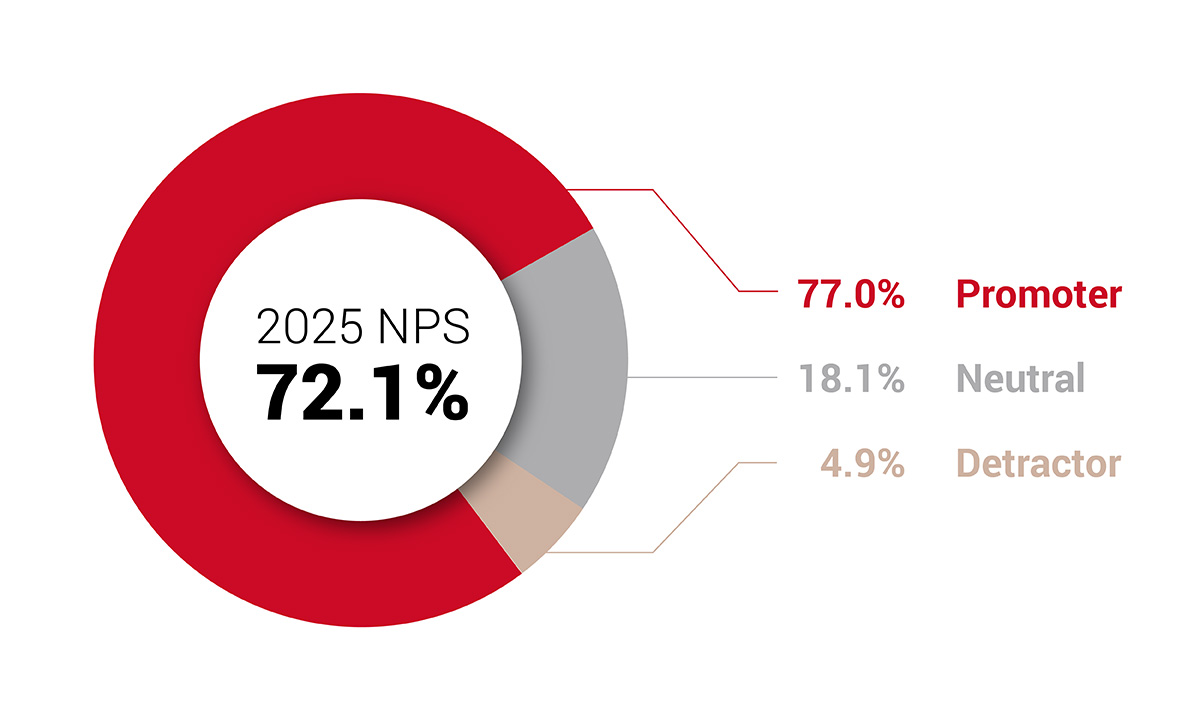
ARUP Laboratories’ 2025 Client Satisfaction Survey shows strong loyalty and excellent service ratings, which reflect continued improvement driven by client feedback.
Kanako Yumioka’s journey from Housekeeping to Specimen Processing shows how mentorship, support, and a “warmful” culture help employees grow and find their path at ARUP Laboratories.
The ARUP Consult Colorectal Cancer topic highlights updated screening recommendations and guidance to help clinicians choose appropriate tests for early detection.
ARUP research highlights include cytogenetic abnormalities of pediatric B-cell acute lymphoblastic leukemia, pharmacogenomic testing, and the influence of diet on biochemical genetic testing.
Tracy George, MD, is a champion for patients with rare and undiagnosed conditions and will speak about ARUP’s innovative efforts that support the rare disease community.
ARUP’s Innovation Central Laboratory will serve as an ecosystem for validating technologies and accelerating next generation diagnostics from concept to commercialization.
Healthcare Advisory Services Vice President Sandy Richman will present strategies for hospital laboratories to launch or grow successful outreach programs and become strategic health system partners.
Ryan Metcalf, MD, CQA(ASQ), has been named to ABPath’s Test Development and Advisory Committee for Blood Banking and Transfusion Medicine, a recognition of his expertise and leadership in the field.
ARUP scientists authored more than 130 peer-reviewed publications and presented at over 100 conferences in 2025, highlighting a year of research, innovation, and progress in diagnostic medicine.