More than a laboratory.
A trusted partner for patient healthcare.
We are ARUP Laboratories, a national, academic, and nonprofit reference laboratory committed to innovating, improving continuously, and helping our clients succeed while serving their patients. We know that every specimen we test represents a patient, a person who matters.
Every test is
Get the Latest From ARUP
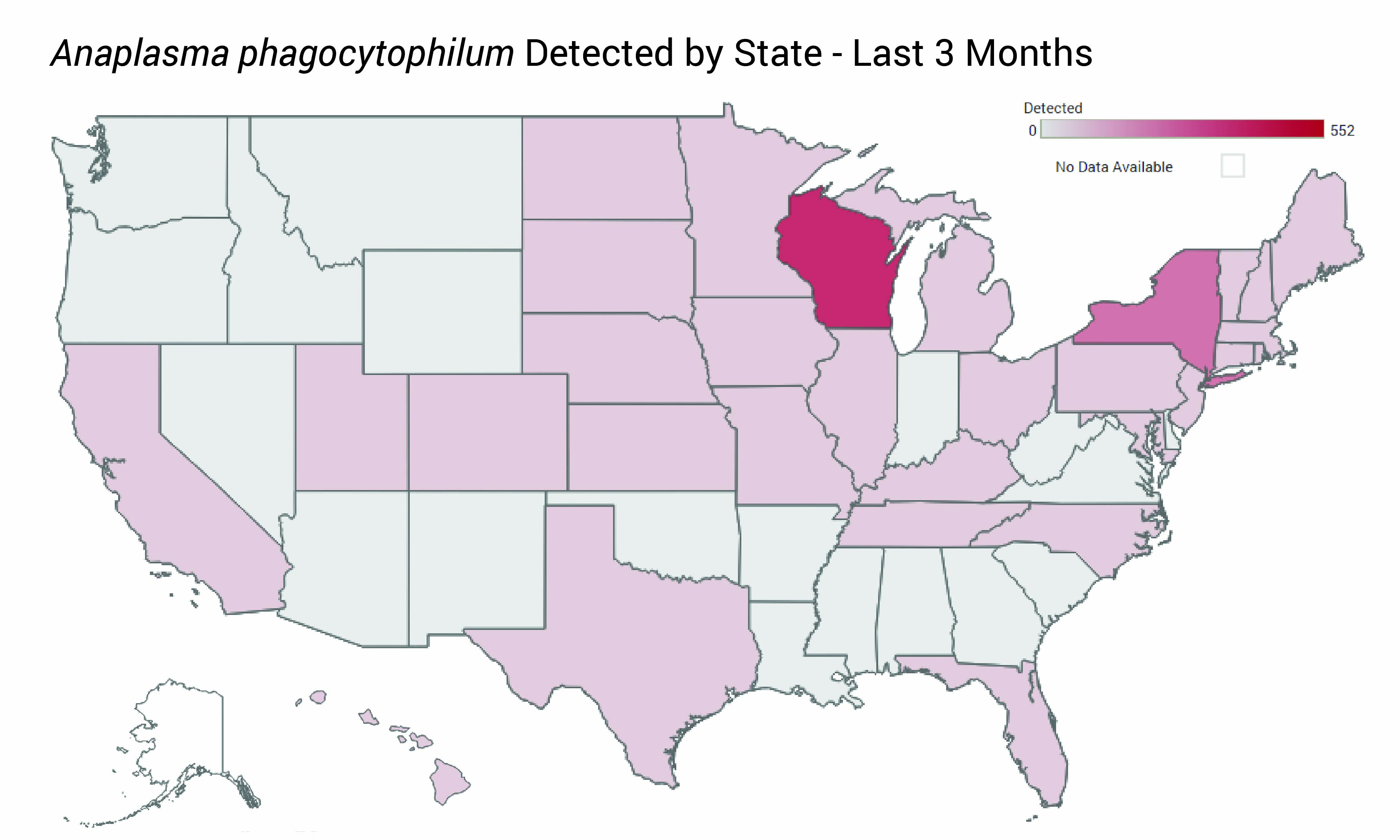
ARUP Launches National Infectious Disease Test Positivity Trends Dashboard
The first national, multipathogen dashboard provides weekly, deidentified test result positivity trends that may help clinical laboratories, medical directors, and clinicians detect unusual patterns, seasonal shifts, and emerging infectious disease activity.
The dashboard features:
- Maps and charts drawn from ARUP’s test results and refreshed weekly
- Pathogen-specific pages accessible through an expandable navigation menu
- “A Key Information” box to call out specific data points