|
April 29, 2026 |
Two ARUP Medical Directors Receive Career Achievement Awards The Pan American Society for Clinical Virology (PASCV) has honored two ARUP experts in clinical virology and public health, recognizing their leadership in advancing infectious diseases testing. |
The Exception Handling team celebrates the winter holidays and keeps their glitter beard tradition alive. |
April 23, 2026 |
Lab Spotlights: Behind the Scenes of Exceptional Patient Care An inside look at three ARUP labs—Mass Spec 1, Exception Handling, and Immunohistochemistry—showing how teamwork, innovation, and behind-the-scenes dedication support accurate testing and care. |
Claire Adam, SPHR, ARUP Human Resources (HR) senior business partner, accepts the Best of BioHive Returnship Champion of the Year Award, April 16, 2026. Pictured in the background, from left to right, Kristine Perez, HR generalist and former returner, Bonnie Stray, supervisor, Corporate Marketing and Public Relations, Rachel Lovado, HR senior business partner, Janani Sankar, research associate and former returner. |
April 20, 2026 | ARUP’s award-winning returnship program helps experienced professionals relaunch their careers while strengthening the life sciences workforce. |
|
April 16, 2026 | Join ARUP in celebrating Lab Week with stories that highlight the many ways laboratory professionals go above and beyond to provide excellent patient care. |
Visit us at booth #4313 to discover intentional laboratory testing for clinically meaningful insights. |
April 8, 2026 |
Discover, Engage, and Explore With ARUP Laboratories at AAN 2026 Connect with ARUP experts to learn about our comprehensive neurology testing suites and how intentional lab testing can support clinically meaningful insights across a range of neurologic conditions. |
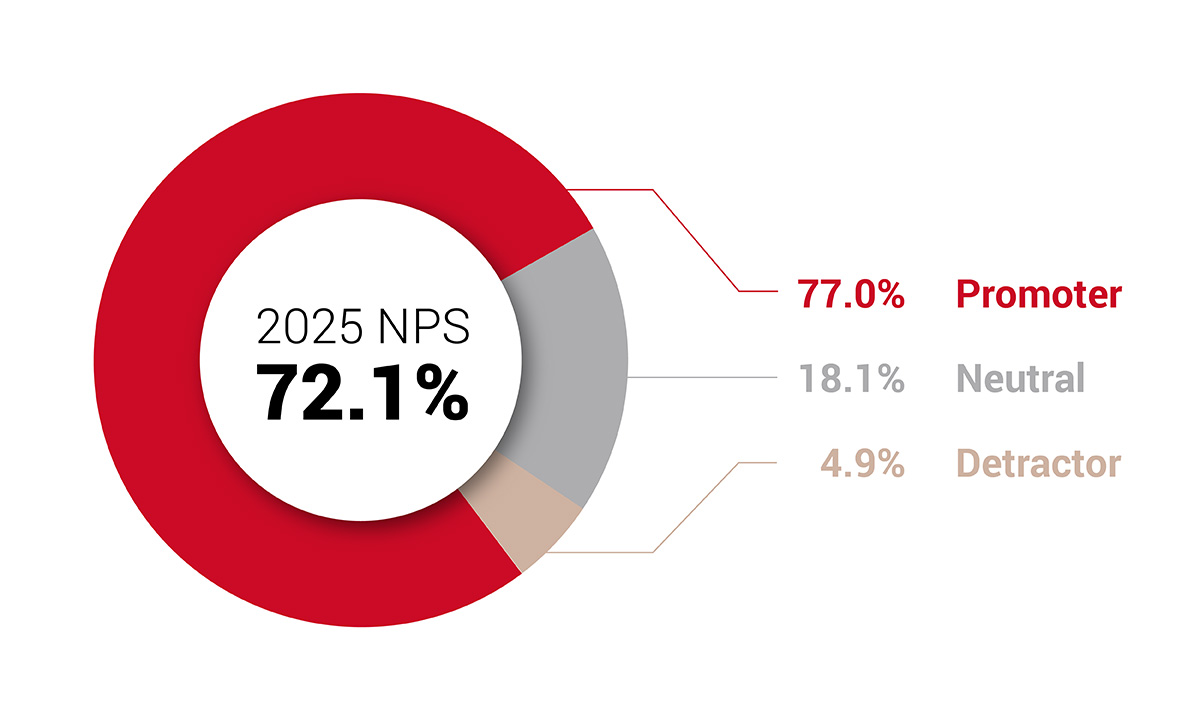
Survey respondents gave ARUP a net promotor score of 72.1%, an increase of over two points from the previous year and placed ARUP into the “excellent” range. |
April 6, 2026 |
ARUP Clients Rate Customer Service High for the Third Consecutive Year in 2025 Satisfaction Survey ARUP Laboratories’ 2025 Client Satisfaction Survey shows strong loyalty and excellent service ratings, which reflect continued improvement driven by client feedback. |