Healthcare Advisory Services Vice President Sandy Richman will present strategies for hospital laboratories to launch or grow successful outreach programs and become strategic health system partners.
Tracy George, MD, ARUP chief scientific officer and Innovation Business Unit president, will help advance life sciences innovation and strengthen Utah’s healthcare ecosystem with BioUtah.
Deploying artificial intelligence (AI) solutions in the lab brings several challenges. In this white paper, ARUP experts offer actionable advice for determining whether AI is right for your lab.
At upcoming conferences, ARUP experts will highlight a new blood-based biomarker assay for Alzheimer’s disease and share a presentation on building a successful laboratory outreach program.
ARUP will expand its AI-augmented screening tool for the detection of human gastrointestinal parasites to become first and only lab to apply AI to the entire ova and parasite testing process.
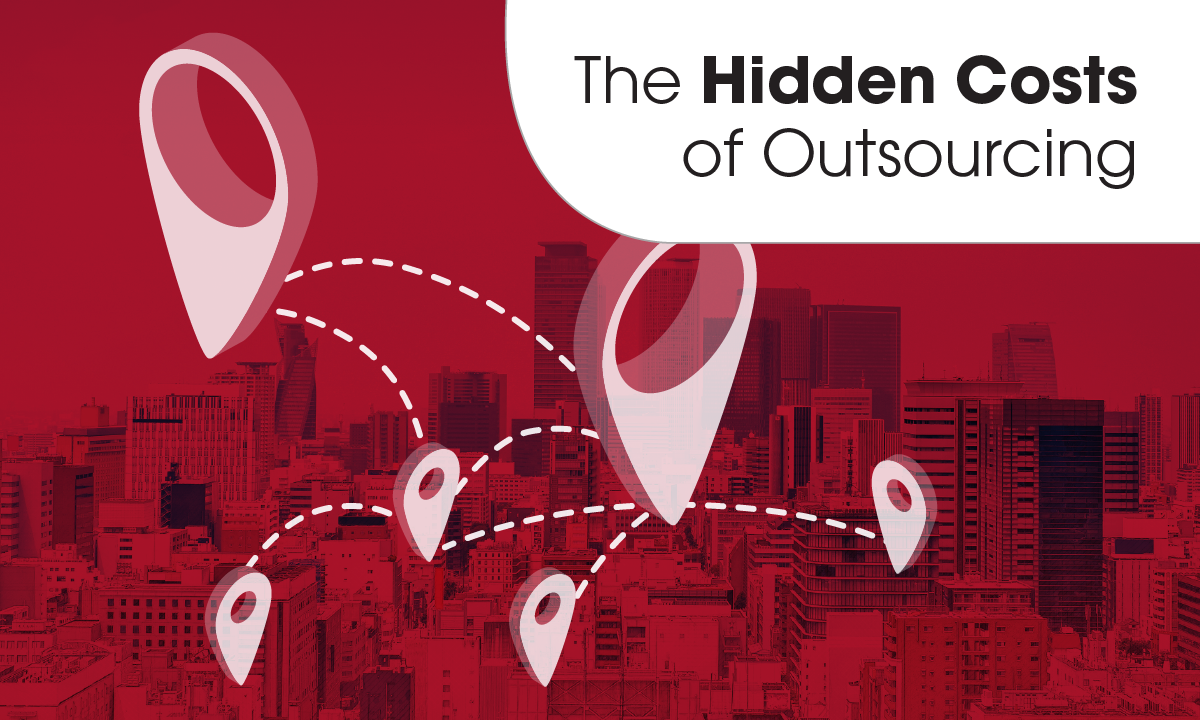
A new ARUP white paper highlights the potential indirect costs of outsourcing laboratories and suggests ways for clinical laboratories to demonstrate their value to the health system.
This edition features interviews with company leaders describing ARUP’s approaches to comply with the FDA’s rule and a video about how ARUP helped a family on a rare disease diagnostic journey.
FDA leaders answered questions on artificial intelligence (AI), international regulations, and representation in clinical trials during the Utah Life Sciences Summit.
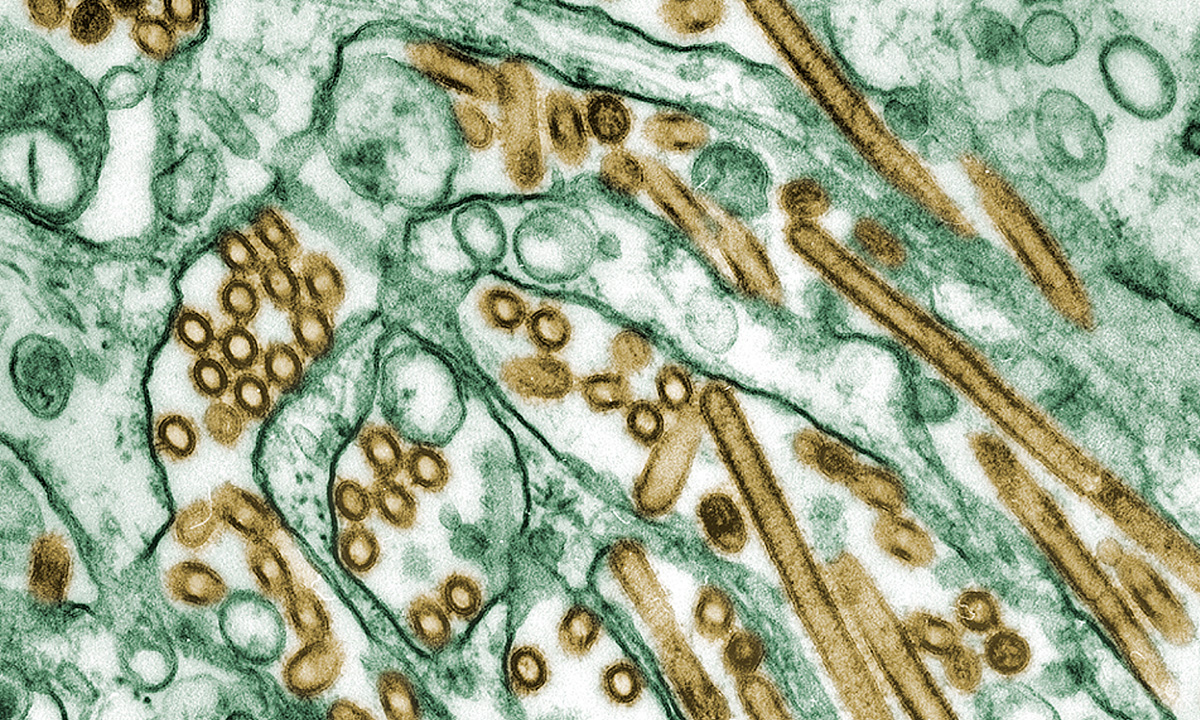
ARUP has been awarded a CDC contract for avian influenza A (H5N1) test development, which recognizes our expertise and experience with assay development and our desire to serve public health needs.
The lone star tick, which can transmit infectious diseases and trigger a serious red meat allergy, is spreading into new areas. ARUP Consult offers guidance to help diagnose tickborne illnesses.
ARUP’s declaration amplifies the lawsuit’s claims that the FDA does not have legal authority over laboratory testing services and that regulating these services is unreasonable.
ARUP Laboratories is committed to helping clients and the clinical laboratory community navigate regulatory changes and is offering current resources and a new webinar.