Foodborne illness can take hold quickly and often presents with acute gastrointestinal (GI) symptoms. |
June 4, 2026 |
ARUP Launches Bacterial Gastrointestinal Panels for Rapid Results and Faster Reporting ARUP has launched multiplex polymerase chain reaction bacterial gastrointestinal panels that detect common pathogens with faster turnaround times to support rapid diagnosis and public health response. |
ARUP experts will share their latest research, including a deep dive on arthropods, drug resistance in mycobacterium, and same-shift diagnostic testing. |
June 1, 2026 |
From Arthropods to Antimicrobial Resistance: ARUP at ASM Microbe 2026 Join ARUP experts at the American Society for Microbiology (ASM) Microbe 2026 conference for their latest research presentations, topic deep dives, and more. |
From left to right, Toni Pollock, director of PharmaDx; Margaret Coppin, HT(ASCP), chief quality officer; Tracy George, MD, chief scientific officer and president of ARUP’s Innovation Business Unit; Sarah Teofilo, chief information officer; Erica Clyde, MBA, PMi-PMP®, MB(ASCP)CM, director of the ARUP Institute for Research and Innovation in Diagnostic and Precision Medicine™; and Ligaya Kuiken, MBA, MLS(ASCP)CM, MB(ASCP)CM, ARUP Human Resources education program coordinator, celebrate George’s selection as one of the 2026 Top 10 Influential Women by Utah Business magazine at a luncheon honoring the Women To Watch nominees. |
May 21, 2026 |
Tracy George, MD, Named One of the Top 10 Influential Women by Utah Business Magazine Tracy George, MD, has been named one of the Top 10 Influential Women by Utah Business magazine, recognizing her leadership in advancing diagnostic innovation, science, and patient care. |
ARUP Laboratories is sponsoring the American Initiative in Mast Cell Diseases Physician and Investigator Conference, and our experts look forward to connecting with attendees at the meeting. |
May 14, 2026 |
Join ARUP Laboratories at the American Initiative in Mast Cell Diseases Conference ARUP experts will present and be available to connect with attendees at the conference in Ann Arbor, Michigan, May 16 and 17. |
Misty Segrest was diagnosed with indolent systemic mastocytosis after a decade-long search for relief from itchy, red bumps on her skin. The diagnosis and treatment brought both relief and renewed quality of life. |
May 14, 2026 |
A Decade Without Answers: The Cancer Diagnosis That Changed Her Life for the Better After years of itchy, red bumps that made her skin feel as if ants were crawling across it, Misty Segrest has her life back—thanks in part to ultrasensitive laboratory testing and targeted treatment. |
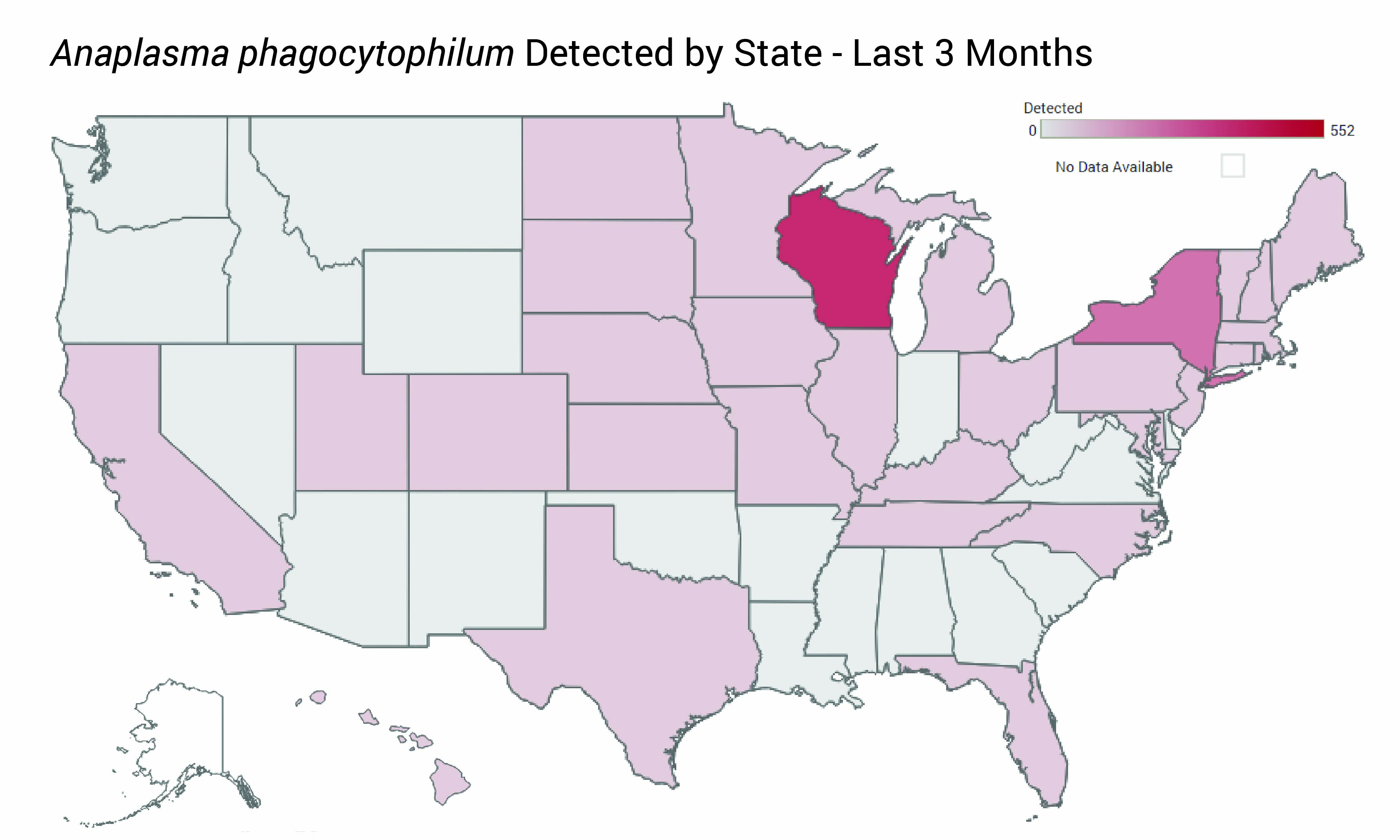
ARUP Laboratories has launched the first national, multipathogen Infectious Disease Test Positivity Trends Dashboard, which provides deidentified test result positivity trends that may help clinical laboratories and medical directors identify unusual patterns, seasonal shifts, and emerging infectious disease activity sooner. |
May 13, 2026 |
ARUP Laboratories Launches National Infectious Disease Test Positivity Trends Dashboard ARUP’s new online dashboard offers clinicians and laboratories unprecedented visibility into pathogen activity through positive test result trends derived from deidentified data. |